Eltrombopag (Revolade, Promacta) is indicated as first-line therapy in patients with chronic immune / idiopathic thrombocytopenia who have failed first-line therapy with glucocorticoids, intravenous immunoglobulins and have low platelets following splenectomy. It has also been studied in patients with chemotherapy-induced thrombocytopenia and thrombocytopenia associated with chronic lymphocytic leukemia (CLL).
Eltrombopag (Promacta) for the treatment of thrombocytopenia secondary to chronic hepatitis C:
Eltrombopag has been studied in patients with thrombocytopenia associated with chronic hepatitis C. The overall response has been estimated to vary between 75 - 95%. The ENABLE 1 and ENABLE 2 studies demonstrated an elevation in the platelet counts resulting in more patients to receive interferon therapy.
The sustained virological response was seen more in patients who received eltrombopag, however, hepatic decompensation was more common in the intervention group. Another similar study demonstrated an overall response (a platelet count of more than 100,000/ul) ranging from 57 - 95%, allowing more patients to complete the 12 weeks treatment period.
Eltrombopag (Promacta) for the treatment of ITP (immune thrombocytopenic purpura):
It has been studied in patients with severe thrombocytopenia secondary to chronic ITP. Patients who were on stable doses of immunosuppressive agents for at least one month and had platelet counts of less than 30000/ul were enrolled. The study showed that eltrombopag when administered at a dose of 50 and 75 mg daily, increased the platelet counts to more than 50000/ul in 80% of the patients in two weeks.
It has also been studied in patients with ITP as repeated cycles of treatment for 6 weeks followed by a 4-week drug-free period. Intermittent dosing may be specifically useful in patients prior to surgery and those who present with active bleeding. Eltrombopag may be used as a second-line therapy in immune thrombocytopenia in patients who do not respond to steroids prior to splenectomy or in patients with contraindications to surgery.
Eltrombopag (Promacta) for the treatment of aplastic anemia:
Eltrombopag approved by the FDA for the treatment of aplastic anemia in patients who fail to respond to immunosuppression. A study was carried out in patients with refractory aplastic anemia who were transfusion-dependent. Patients were given increasing doses of eltrombopag (to the maximum dose of 150 mg per day) for 12 weeks.
Significant responses in all cell lines were noted (platelet, erythroid, and neutrophil lineages) in 11 of 25 patients at 12 weeks. Normalization of bone marrow cellularity and trilineage hematopoiesis was observed in patients who continued to receive eltrombopag. 9 out of 25 patients remained transfusion independent. two patients who did not respond to the treatment had monosomy 7.
Progression to myelodysplasia and clonal hematopoiesis was not seen. Eltrombopag has also been studied with concomitant immunosuppressants in patients with severe aplastic anemia who were 12 years of age or older. Patients received eltrombopag in addition to anti-thymocyte globulin (ATG - ATGAM) and cyclosporin. More than 1/3 rd of the patients had a complete hematologic response which was defined as:
- hemoglobin of more than 10 gm/dl
- Platelet counts of more than 100,000/ul
- Absolute neutrophil counts of more than 1000/ul
The overall response rate was 94% and a complete response was seen in 58% of the patients.
Eltrombopag (Promacta) for the treatment of ITP in children:
The PETIT2 trial evaluated the safety and efficacy of eltrombopag in children aged 1 - 17 years of age. The study was carried out in 38 centers (12 countries). All patients had chronic ITP with a platelet count of less than 30,000/ul. The study concluded that 40% of the children had a sustained platelet response of more than 50,000/ul.
It was very well tolerated with few adverse events like elevated liver enzymes. It is currently approved for use in children older than one year with chronic ITP who fail to respond to first-line therapy. Eltrombopag has also been used in children with aplastic anemia who develop thrombocytopenia after HSCT.
Eltrombopag vs Romiplostim:
Both these drugs increase platelet counts by acting on the thrombopoietin receptors. The following table is a comparison of the two drugs: Comparison of Eltrombopag and romiplostim in children with chronic ITP:
Eltrombopag (ELT) |
Romiplostim (ROM) |
|
| Mechanism | Non-peptide TPO receptor agonist that binds to a transmembrane site of TPO receptor | Peptide mimetic TPO receptor agonist that binds to the extracellular TPO receptor |
| FDA approval | ITP (Children and adults) Aplastic anemia | ITP only |
| Mode of administration | oral | subcutaneous |
| Platelet response | Increase platelets ( Overall platelet response between ELT and ROM was not different) | Increase platelets ( Overall platelet response between ELT and ROM was not different) |
| Side effects | Cough more frequent when compared to ROM | Except for cough, other side effects in both the drugs are the same. |
| Durable platelet response | No significant difference | No significant difference |
| Bleeding risk | Low (compared to ROM) | High (compared to ELT) |
| Cost of one dose | (Cost of each tablet) 12.5 mg: $197.06 25 mg: $197.06 50 mg: $356.61 75 mg: $534.92 | (Cost of each injection) 125 mcg: $1,115.15 250 mcg: $2,230.30 500 mcg: $4,460.59 |
| Onset of action | 1 – 2 weeks | 4 – 9 days |
| Peak platelet response | 2 – 4 weeks | 12 – 16 days |
| Half-life | 21 – 32 hours | 3.5 days |
| Brand names | Promacta Rebozet Revolade Versapenia | Nplate Romiplate |
| Food interactions | Take it 1 hour before or 2 hours after a meal, Take 2 hours before or 4 hours after foods rich in calcium | none |
Adult dose:
Eltrombopag dose for ITP (Immune thrombocytopenic purpura):
Oral Tablets: Initial: 50 mg once daily (25 mg once daily for patients of East-Asian ethnicity). The dose should be titrated based on platelet response.
The maximum dose is 75 mg once daily.
How to adjust the dose while on therapy:
-
Platelet counts of less than 50,000/mm 3 while on treatment for 2 weeks or more:
- Increase daily dose by 25 mg (if taking 12.5 mg once daily, increase dose to 25 mg once daily prior to increasing the dose amount by 25 mg daily); maximum dose is 75 mg once daily
-
Platelet counts of 200,000/mm 3 or more and 400,000/mm 3 or less at any time:
- Reduce the daily dose by 25 mg; reassess in 2 weeks
-
Platelet counts of more than 400,000/mm 3 :
- Withhold dose; assess platelet count twice weekly; when platelet count <150,000/mm 3, resume with the daily dose reduced by 25 mg (if taking 25 mg once daily, resume with 12.5 mg once daily)
-
Platelet count of more than 400,000/mm 3 after 2 weeks at the lowest dose:
- Discontinue treatment
Note: Use the lowest dose to achieve and maintain platelet count ≥50,000/mm 3 as needed to reduce the risk of bleeding.
Discontinue if platelet count does not respond to a level that avoids clinically important bleeding after 4 weeks at the maximum daily dose of 75 mg.
Eltrombopag dose in chronic hepatitis C-associated thrombocytopenia:
Oral: Initiate treatment with 25 mg once daily. Adjust the dose according to treatment response.
(Maximum dose: 100 mg once daily)
Dosage adjustment based on platelet response:
-
Platelet count less than 50,000/mm 3 after at least 2 weeks of therapy:
- Increase daily dose by 25 mg every 2 weeks; maximum dose: 100 mg once daily
-
Platelet count of more than 200,000/mm 3 and less than 400,000/mm 3 at any time:
- Reduce daily dose by 25 mg and reassess in 2 weeks' time.
-
Platelet counts of more than 400,000/mm 3:
- Withhold the dose and assess platelet count twice weekly.
- When platelets count are less than 150,000/mm3, resume with the daily dose reduced by 25 mg (if taking 25 mg once daily, resume with 12.5 mg once daily)
-
Platelet counts of more than 400,000/mm 3 after 2 weeks at the lowest dose:
- Discontinue treatment
Note: Use the lowest dose to achieve the target platelet count necessary to initiate antiviral therapy or to avoid dose reductions of peginterferon during antiviral therapy. Discontinue when antiviral therapy is stopped. 
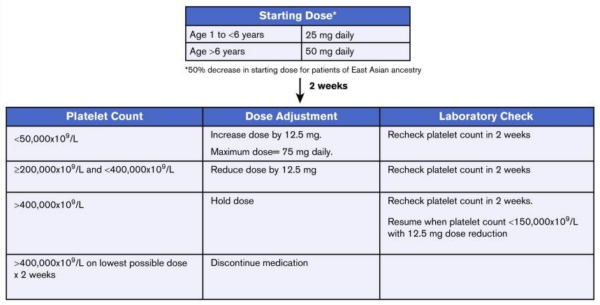
Dose in children:
Eltrombopag dose for ITP in children:
The starting dose is 50 mg per daily for patients aged 6 years or older and 25 mg per day for patients aged 1 to younger than 6 years. Once initiated, eltrombopag dosing should be adjusted to achieve a platelet count goal of 50 × 109/L, not to exceed 200 × 109/L. The dose should not be titrated to achieve normal platelet counts. The dose can be escalated every 2 weeks in increments of 12.5 mg to achieve the goal platelet range.
The maximum dose of eltrombopag for the treatment of ITP is 75 mg per day
Eltrombopag Pregnancy Risk Factor C
The manufacturer recommends avoiding eltrombopag in pregnancy and during breastfeeding.
Eltrombopag dose in kidney disease:
- Dose adjustment not necessary in renal disease
Dose in liver disease:
Adjustment dose in hepatic impairment prior to initiating treatment:
-
Hepatic impairment of any degree including mild, moderate, and severe impairment (Child-Pugh classes A, B, or C):
- Initiate with 25 mg once daily
-
Patients of East-Asian ethnicity with hepatic impairment (Child-Pugh classes A, B, or C):
- Initiate therapy with 12.5 mg once daily
Adjustment dose in hepatic impairment during treatment:
Eltrombopag use has been associated with hepatotoxicity. Baseline Liver function tests should be performed.
-
Eltrombopag should be discontinued if:
- The ALT is 3 times the upper limit of normal (ULN) or more in patients with normal hepatic function, or
- More than 3 times the baseline in those with pre-existing transaminase elevations and which are progressive, or
- Persistent (4 weeks or more), or
- Accompanied by increased direct bilirubin, or
- Accompanied by clinical signs of liver injury or evidence of hepatic decompensation.
Reinitiation with a low dose and strict monitoring may be considered in some patients. Patients who develop hepatotoxicity with a rechallenge should permanently discontinue eltrombopag.
Eltrombopag side effects (Common)
- Abdominal pain
- Alopecia
- Arthralgias and myalgias
- Bone pain
- Cataract & dry eyes
- Nausea, Constipation or Diarrhea
- Fatigue & headache
- Insomnia, paraesthesias
- Edema, pruritis, and a rash
Eltrombopag side effects (uncommon)
- Anemia,
- anorexia,
- anxiety,
- myocardial infarction,
- cytopenias,
- cholestasis,
- cough,
- deep vein thrombosis,
- depression,
- dizziness,
- ecchymosis,
- eosinophilia,
- flushing,
- gout,
- hemolysis,
- hemorrhoids,
- hepatitis,
- hypertension,
- migraine,
- nocturia,
- palpitation,
- QT-interval prolongation,
- renal failure,
- distaste,
- tremors,
- vertigo,
- weight gain,
- urinary tract infections and
- rectosigmoid cancers.
Eltrombopag contraindications and cautions
- The manufacturer's labeling does not contain any contraindications.
Warning and precautions
- Bone marrow fibrosis:
- It is important to monitor patients for any new-onset cytopenias.
- Eltrombopag has been shown to increase the formation of reticulin and cause fibrosis. In such cases, a repeated bone marrow biopsy may be warranted.
- Cataract formation:
- Eye disease can occur in patients taking eltrombopag, especially those who are older or take high-dose steroids for a long time.
- Hepatotoxicity: [U.S. Boxed Warning]:
- Hepatotoxicity has been linked to Eltrombopag usage. It is important to perform baseline liver function tests.
- If the ALT exceeds 3 times the upper limit normal (ULN), in patients with normal hepatic function or greater, Eltrombopag should not be used.
- Patients with pre-existing elevations of transaminases that are progressive or more than three times the baseline will have their levels exceed 3x.
- Persistent (>=4 Weeks), or
- Increased direct bilirubin or clinical signs of liver injury, or evidence of hepatic dysfunction.
- Some patients may need to be reintroduced at a lower dose.
- Patients who are not able to monitor their progress should be advised.
- Patients with hepatotoxicity after a rechallenge should discontinue using eltrombopag.
- Hepatotoxicity has been linked to Eltrombopag usage. It is important to perform baseline liver function tests.
- Malignancy/tumorigenicity:
- The risk of developing hematologic malignancies may be increased by stimulation of the cell surface receptors for thrombopoietin.
- Thromboembolism:
- Venous thromboembolism may occur with thrombocytosis especially in patients with risk factors like protein C, S deficiency, Factor V Leiden mutations, and anti-Thrombin III deficiency.
- Portal vein thrombosis is a condition that occurs in patients with chronic hepatitis C. This can be caused by treatment for thrombocytopenia using eltrombopag.
- Chronic Hepatitis C Infection: [U.S.Boxed Warning]
- Patients with chronic hepatitis A may be at greater risk from hepatic decompensation if Eltrombopag is used in combination of interferon and Ribavirin.
Eltrombopag: Drug Interaction
|
Risk Factor C (Monitor therapy) |
|
|
BCRP/ABCG2 Substrates |
Eltrombopag may increase the serum concentration of BCRP/ABCG2 Substrates. |
|
CycloSPORINE (Systemic) |
May decrease the serum concentration of Eltrombopag. |
|
Deferiprone |
UGT1A6 Inhibitors may increase the serum concentration of Deferiprone. |
|
OATP1B1/1B3 (SLCO1B1/1B3) Substrates |
Eltrombopag may increase the serum concentration of OATP1B1/1B3 (SLCO1B1/1B3) Substrates. |
|
Talazoparib |
BCRP/ABCG2 Inhibitors may increase the serum concentration of Talazoparib. |
|
Risk Factor D (Consider therapy modification) |
|
|
Cladribine |
BCRP/ABCG2 Inhibitors may increase the serum concentration of Cladribine. Management: Avoid concomitant use of BCRP inhibitors during the 4 to 5 day oral cladribine treatment cycles whenever possible. If combined, consider dose reduction of the BCRP inhibitor and separation in the timing of administration. |
|
Eluxadoline |
Eltrombopag may increase the serum concentration of Eluxadoline. Management: Decrease the eluxadoline dose to 75 mg twice daily if combined with eltrombopag and monitor patients for increased eluxadoline effects/toxicities. |
|
Polyvalent Cation Containing Products |
May decrease the serum concentration of Eltrombopag. Management: Administer eltrombopag at least 2 hours before or 4 hours after oral administration of any polyvalent cation containing product. |
|
Rosuvastatin |
Eltrombopag may increase the serum concentration of Rosuvastatin. Management: Consideration a preventive 50% reduction in rosuvastatin adult dose when starting this combination; Canadian labeling recommends limiting rosuvastatin to a maximum of 20 mg/day. |
|
Risk Factor X (Avoid combination) |
|
|
Asunaprevir |
OATP1B1/1B3 (SLCO1B1/1B3) Inhibitors may increase the serum concentration of Asunaprevir. |
|
Elagolix |
OATP1B1/1B3 (SLCO1B1/1B3) Inhibitors may increase the serum concentration of Elagolix. |
|
Grazoprevir |
OATP1B1/1B3 (SLCO1B1/1B3) Inhibitors may increase the serum concentration of Grazoprevir. |
|
PAZOPanib |
BCRP/ABCG2 Inhibitors may increase the serum concentration of PAZOPanib. |
|
Revefenacin |
OATP1B1/1B3 (SLCO1B1/1B3) Inhibitors may increase serum concentrations of the active metabolite(s) of Revefenacin. |
|
Topotecan |
BCRP/ABCG2 Inhibitors may increase the serum concentration of Topotecan. |
|
Voxilaprevir |
OATP1B1/1B3 (SLCO1B1/1B3) Inhibitors may increase the serum concentration of Voxilaprevir. |
How and when to monitor?
- Monitor liver function before treatment, every two weeks when adjusting the dose, and monthly thereafter.
- Regular ophthalmological examinations for cataract formation recommended.
- Monitor full blood count including platelet count and peripheral blood smears every week during treatment until a stable platelet count is reached (50,000/ul or more for at least 4 weeks), then monthly thereafter.
How and when to take eltrombopag (Revolade)?
- Administer on an empty stomach, 1 hour before or 2 hours after a meal.
- Food, especially dairy products, may decrease the absorption of eltrombopag.
- Allow at least 4 hours between dosing of eltrombopag and polyvalent cation intake (eg, dairy products, calcium-rich foods, multivitamins with minerals).
- Do not administer concurrently with antacids, foods high in calcium, or minerals (eg, iron, calcium, aluminum, magnesium, selenium, zinc); separate by at least 4 hours.
- Do not administer more than one dose within 24 hours.
Eltrombopag mechanism of Action:
- Eltrombopag binds with the Thrombopoietin receptors (TPO) and activates them.
- This increases proliferation and differentiation in the marrow progenitor cell cells.
The expected increase in platelet counts: Within 1-2 weeks
Peak platelet count increase: 14-16 days
Duration: Platelets return to baseline: 1-2 weeks after the last dose
Protein binding: >99%
Metabolism: Extensive hepatic metabolism
Bioavailability: about 52%
Half-life elimination: about 21-32 hours in healthy individuals; 26-35 hours in patients with ITP
Time to peak plasma concentration: 2-6 hours
Excretion: Mainly via feces
International Brands of Eltrombopag
- Revolade
- Rebozet
- Promacta
Eltrombopag brands in Pakistan and price:
Revolade (GlaxoSmithKline)
-
Tablets 25 mg:
- 28 tablets: Rs. 75600
-
Tablets 50 mg
- 28 tablets: Rs. 126000